.png)
In the periodic table, we can see that Beryllium has Z = 4. Example 1īeryllium-9 absorbs an alpha particle during a nuclear reaction, producing an unknown nucleus and a neutron. If we use an analogy with linear momentum, nuclear reactions are analogue to collisions (both elastic and inelastic) while decay processes are analogue to explosions, as the energy stored in atomic nuclei is activated when a radioactive decay takes place, similar to energy stored in chemical form that is activated during explosions. Likewise, when the total mass of system increases after the reaction, the energy is absorbed by the system ( Q < 0).ĭo not confuse nuclear reactions with radioactive decay processes. Thus, based on mass-energy equivalence, when Q > 0, then the total mass of reaction products is smaller than the mass of target nuclei plus that of hitting particle. It represents the energy released or absorbed during a nuclear reaction. Where X is the target nucleus, a is the hitting particle, Y is the daughter (hit) nucleus and b is the particle/s produced during the reaction.Īnother important quantity to be considered during a nuclear reaction is the energy of reaction, Q. Schematically, a nuclear reaction is written as: The hitting particle may be a proton, neutron, gamma ray or an entire atomic nucleus. As a result, a new (daughter) nucleus is obtained, where some energy (and nuclear particles) is/are released or absorbed, depending on the type of nuclear reaction. Thus, in a nuclear reaction, a target (parent) nucleus is hit by another nuclear particle. Nuclear reactions are apparently similar to chemical reactions (where we have to consider the amount of reactants and product based on the law of mass and energy conservation). The particles involved in nuclear reactions include atomic nuclei and other particles either involved in the process or produced during them, as well as the corresponding radiation emitted. The term " nuclear reactions" refers to a special category of matter-energy interaction that includes all processes in which two or more objects interact with each other through nuclear forces. In addition - among many other things - we will explain the thermonuclear reactions, as a special category of nuclear reactions, as they have a wide range of use in daily life. We will explain various types of nuclear reactions as well as how nuclear energy (which is produced in abundance during nuclear reactions) is used in industry. In this article, we will discuss about nuclear reactions - a prerequisite to produce nuclear energy. What is nuclear energy used for? Give some examples. Have you ever heard about nuclear reactors? How do they work?ĭo you think it is possible to merge two atomic nuclei at home conditions? What about to split an atomic nucleus in two smaller nuclei? Why? What are the advantages and disadvantages of nuclear energy use?. 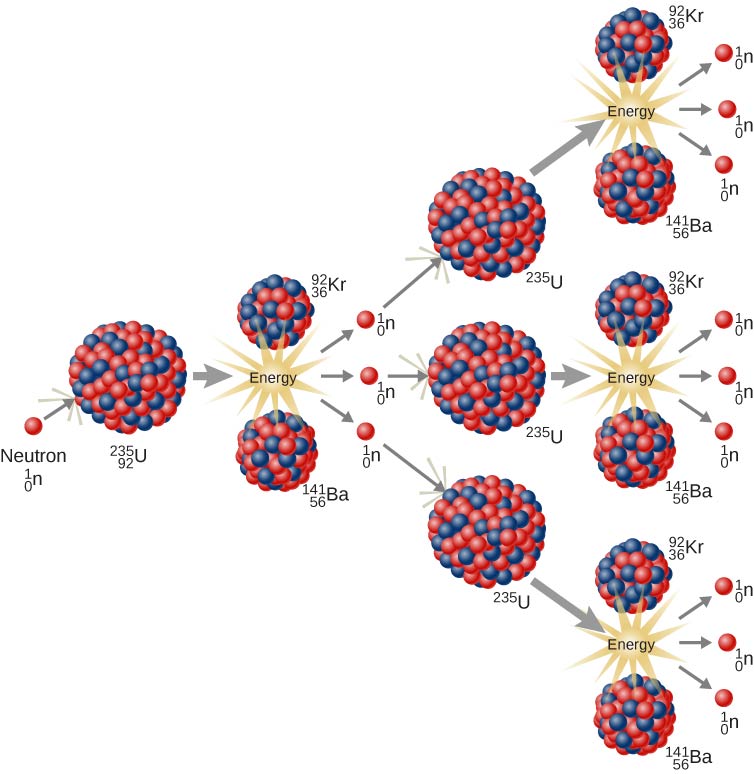
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
